User:Mr. Ibrahem/Travoprost
 | |
| Clinical data | |
|---|---|
| Trade names | Travatan, Izba, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602027 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Topical (eye drops) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Activation by ester hydrolysis, deactivation by beta oxidation, OH-oxidation, double bond reduction |
| Onset of action | 2 hours |
| Elimination half-life | 1.5 hours (in aqueous fluid) 45 minutes (terminal) |
| Duration of action | ≥ 24 hours |
| Excretion | Mainly via kidney |
| Identifiers | |
| |
| Chemical and physical data | |
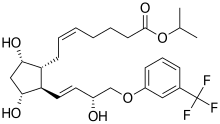
| Formula | C26H35F3O6 |
| Molar mass | 500.555 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Travoprost, sold under the brand name Travatan among others, is medications used to treat high pressure inside the eye including glaucoma.[3] Specifically it is used for open angle glaucoma when other agents are not sufficient.[4][3] It is used as an eye drop.[3] Effects generally occur within 2 hours.[3]
Common side effects include red eyes, blurry vision, eye pain, dry eyes, and change in color of the eyes.[3][4] Other significant side effects may include cataracts.[4] Use during pregnancy or breastfeeding is generally not recommended.[4] It is a prostaglandin analog and works by increasing the outflow of aqueous fluid from the eyes.[3]
Travoprost was approved for medical use in the United States in 2001.[3] It is available as a generic medication in the United Kingdom.[4] A 2.5 milliliter bottle in the United Kingdom costs the NHS about 7.30 £ as of 2019.[4] In the United States the wholesale cost of this amount is about US$177.[5] In 2017, it was the 212th most commonly prescribed medication in the United States, with more than two million prescriptions.[6][7]
References[edit]
- ^ a b "Travoprost ophthalmic Use During Pregnancy". Drugs.com. 8 October 2019. Retrieved 16 May 2020.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Retrieved 9 September 2020.
- ^ a b c d e f g "Travoprost Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 26 March 2019.
- ^ a b c d e f British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1152. ISBN 9780857113382.
- ^ "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Retrieved 11 April 2020.
- ^ "Travoprost - Drug Usage Statistics". ClinCalc. Retrieved 11 April 2020.
